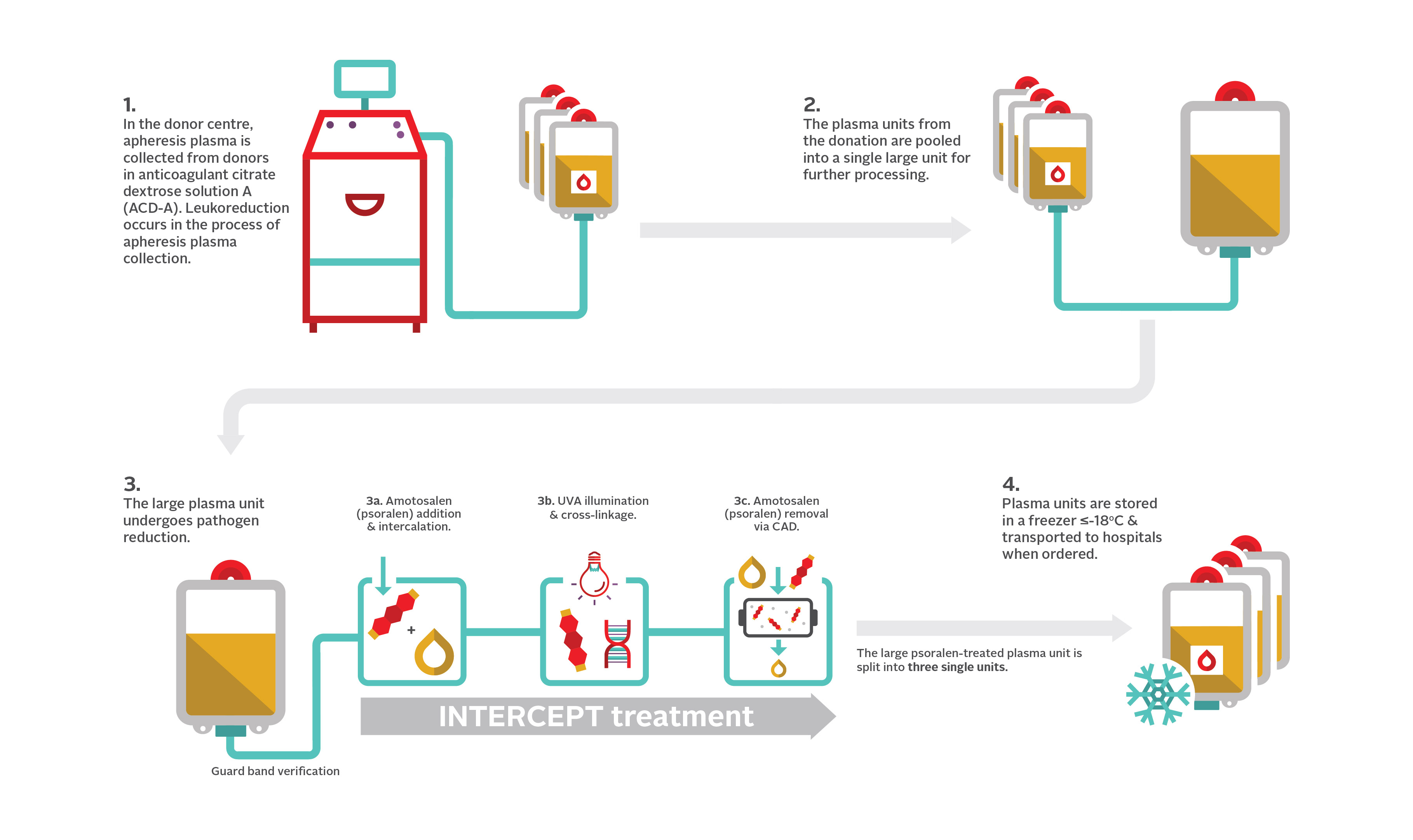
15. How is apheresis frozen plasma, psoralen-treated administered?
The volume of apheresis frozen plasma, psoralen-treated transfused depends on the clinical situation and recipient size. Common dosing is 10–15 mL per kg body weight.
Serial laboratory assays of coagulation function may be of assistance in planning the dose. A standard blood administration set containing a 170–260-micron filter or a filter of equivalent efficacy, approved by Health Canada, must be used for infusion.
No medications or solutions may be added to or infused through the same tubing simultaneously with blood or blood components, unless the solution has been approved for this use by Health Canada or there is documentation available to show that addition of the solution to the blood component involved is safe. Please refer to the Circular of Information for Plasma components for further information.
Transfusion rate is dependent on clinical factors. For more information, refer to the Clinical Guide to Transfusion. All transfusions should be complete within 4 hours of removal from storage. Recipients should be under observation during transfusion with close observation during the first 15 minutes and in accordance with institutional guidelines.
16. What are the indications for apheresis frozen plasma, psoralen-treated?
Apheresis frozen plasma, psoralen-treated shares the same indications as fresh plasma and may be useful in the management of:
- Bleeding patients or patients undergoing invasive procedures with high bleeding risk who require replacement of multiple plasma coagulation factors.
- Patients receiving massive transfusion with clinically significant coagulation abnormalities.
- Patients on warfarin who are bleeding or need to undergo a high bleeding risk invasive procedure before vitamin K could reverse the warfarin effect, and where prothrombin complex concentrate is not available or is contraindicated.5-7
- Patients with selected coagulation factor or with rare specific plasma protein deficiencies for which a more appropriate alternative therapy is not available.
- Preparation of reconstituted whole blood for exchange transfusion in neonates.
- Patients with thrombotic thrombocytopenic purpura (TTP) or hemolytic uremic syndrome (HUS) undergoing plasma exchange.
17. Are there any contraindications in using apheresis frozen plasma, psoralen-treated?
Patients with known anaphylaxis to plasma components should only receive plasma components under appropriate medical supervision. Recipients with known anti-IgA should receive IgA deficient plasma.
Plasma components should not be used to treat hypovolemia.
Apheresis frozen plasma, psoralen-treated should not be used for:
- Patients with a history of hypersensitivity reaction to amotosalen or other psoralens.
- Neonatal patients treated with phototherapy devices that emit a peak energy wavelength less than 425 nm, and/or have a lower bound of the emission bandwidth <375 nm, due to the potential for erythema resulting from interaction between ultraviolet light and amotosalen.
18. How should suspected reactions to apheresis frozen plasma, psoralen-treated be managed?
Management of suspected reactions should follow recommendations for adverse reactions to conventional blood components. See Reporting adverse-transfusion reactions for more information.
Potential adverse events related to a blood transfusion range in severity from minor with no sequelae to life-threatening. All adverse events occurring during a transfusion should be evaluated to determine whether the transfusion can be safely continued/restarted. All adverse events suspected to be related to a transfusion (whether during or after a transfusion) should be reported to your local transfusion service and, when required, the hospital/regional hemovigilance network. Health Canada’s Blood Regulations requires reporting of adverse events that may be attributed to the manufacturing process (e.g., associated with blood component quality) to Canadian Blood Services.8-11
Reporting of suspected cases of transfusion-related infections such as HIV, HCV, HTLV, HBV, West Nile Virus and other transfusion-related infections is described in Chapter 1 of the Clinical Guide to Transfusion and in the publication, A guide to reporting adverse transfusion reactions.
19. What are the benefits of using pathogen-reduced plasma?
Pathogen inactivated plasma has an added layer of protection against bacterial contaminants (gram-positive and gram-negative), viruses (enveloped and non-enveloped), protozoa parasites and white blood cells (leukocytes).
In addition to a reduction in the risk of pathogen transmission, other adverse transfusion reactions also decrease with use of pathogen reduction technology such as solvent detergent plasma. These include a reduction in allergic reactions and febrile non hemolytic transfusion reactions; however, these reductions may reflect pooling of plasma and subsequent dilution of potential allergens or reactants rather than the pathogen-reduction process.
20. Can apheresis frozen plasma, psoralen-treated be used to treat neonatal and pediatric patients?
There are limited data available for the use of pathogen-reduced plasma for transfusion among pediatric and neonatal patients. However, the available hemovigilance data suggests the product is safe and well tolerated in this population.
Preliminary animal studies of pathogen-reduced plasma reported no adverse impacts with transfusion in neonatal rats. Specifically, there were no amotosalen-related adverse effects found in the neonatal rats despite the exposure to amotosalen concentrations being as high as 48 times the standard exposure in adult patients.12
Human clinical trials have similarly reported no or low-grade adverse events among pediatric and neonatal populations. An open label clinical trial conducted in Belgium and France reported that pathogen-reduced plasma transfusion was well tolerated in routine clinical use for children and infants.13 The study assessed 160 children (aged 1–-18 years) and 188 infants (aged < 1 years) from 2007–2011, constituting 11% of the total mixed hematology, medical and surgical patients in the study (n=3, 232). Results for pediatric patients were not reported separately from the adult patients, but overall adverse transfusion reactions were reported in 0.11% (8/7483) of transfusions, with 5 being of Grade 1 severity and 3 serious adverse events; all adverse events occurred in adult patients in this study. There were no reported TRALI events.
Another hemovigilance study assessing the safety profile of pathogen-reduced platelets and pathogen-reduced plasma in 11 countries over an 8-year period reported infrequent transfusion reactions among patients (0.4% of transfusions). The study assessed 440 infants (aged < 1 years) and 355 children (aged 1–18 years), and the pediatric data was not reported separately from adult patients. A subsequent post-marketing hemovigilance study of 3, 179 pathogen-reduced plasma and platelet transfusions in 11 countries found that these components were well tolerated by a pediatric population with only low-grade transfusion reactions reported. The sample included 540 children (aged 1–18 years) and 499 infants (aged < 1 year) with mixed clinical indications for transfusion, including medical (45%), hematology-oncology (34%) and surgical (20%). Three of the 795 pediatric patients experienced a transfusion reaction. Symptoms were characterized by rash (n=2), urticaria (n=1), and chills (n=1) without serious consequences, TRALI, or transfusion associated sepsis.14
From a hemostasis perspective, a single-arm open label study of pathogen-reduced plasma reported good clotting factor kinetics post-transfusion and hemostatic efficacy when used for both prophylactic and therapeutic indications. The study included 34 patients who received 107 transfusions; a portion of the participants were pediatric patients (ages ranged from 3 to 77 years) but data for pediatric patients were not reported separately from adult patients.15
Currently, there is limited safety data for apheresis frozen plasma, psoralen-treated when used for intrauterine transfusion.16
21. What dose of apheresis frozen plasma, psoralen-treated should be prescribed?
The volume of apheresis frozen plasma, psoralen-treated transfused depends on the clinical situation and recipient size. Common dosing is 10–15 mL per kg body weight.
Serial laboratory assays of coagulation function may be of assistance in planning dose.
22. How does apheresis frozen, plasma psoralen-treated compare to solvent detergent plasma and untreated frozen plasma regarding factor levels?
Table 2. Comparison of day 0 post-thaw plasma in vitro quality across plasma types
| Parameter |
Untreated non-O multi-plasma
(n = 15) †
|
INTERCEPT treated non-O
multi-plasma
(n = 33) †‡ |
Octaplasma
(n = 12) § |
Laboratory clinical reference range, 95% CI|| |
|---|
| |
Mean |
SD |
Min |
Max |
Mean |
SD |
Min |
Max |
Mean |
Min |
Max |
|---|
| FVIII (IU/mL)¶ |
1.36 |
0.31 |
0.67 |
1.88 |
0.83 |
0.18 |
0.37 |
1.15 |
1.08** |
0.80 |
1.30 |
0.50-1.49 |
|---|
| FVII (IU/mL) |
1.13 |
0.26 |
0.69 |
1.67 |
0.87 |
0.19 |
0.38 |
1.27 |
1.08 |
0.90 |
1.17 |
0.45-1.33 |
|---|
| FV (IU/mL) |
0.94 |
0.11 |
0.71 |
1.17 |
1.01 |
0.21 |
0.63 |
1.36 |
0.78 |
0.75 |
0.84 |
0.66-1.02 |
|---|
| Fibrinogen (g/L) |
3.09 |
0.51 |
2.22 |
3.76 |
2.53 |
0.56 |
1.82 |
3.68 |
2.50 |
2.40 |
2.60 |
1.70-4.10 |
|---|
| Prothrombin time (s) |
14.00 |
0.50 |
13.20 |
14.80 |
15.10 |
0.8 |
13.90 |
17.50 |
No data |
10.40-12.30 |
|---|
| Protein S activity (IU/mL) |
1.02 |
0.18 |
0.66 |
1.30 |
0.83 |
0.15 |
0.55 |
1.22 |
0.64 |
0.55 |
0.71 |
0.55-1.43 |
|---|
| Alpha-2 antiplasmin (IU/mL) |
1.20 |
0.14 |
1.01 |
1.53 |
1.02 |
0.12 |
0.81 |
1.29 |
0.23 |
0.20 |
0.27 |
0.68-1.26 |
|---|
| ADAMTS-13 (IU/mL) |
0.78 |
0.18 |
0.54 |
1.07 |
0.93 |
0.12 |
0.67 |
1.24 |
1.01 ± 0.08
(mean ± SD) |
40%–130%# |
|---|
†Alpha-2 antiplasmin and ADAMTS-13 were determined using kits designed for research purposes. The other parameters were tested on Stago Coagulation Analyzer. All methods are designed for clinical diagnostic samples; however, we are using them “off label” to measure coagulation related parameters in plasma components (n = 33 for all the parameters).
‡Data were gathered from development data at Canadian Blood Services.
§All parameters taken from product monograph (n = 12),17 except for ADAMTS-13 (n =18) taken from https://www.octaplasma.ca/standardization-of-plasma-protein-levels-in-octaplasma/
||There are currently no defined acceptance criteria listed by CSAZ902-258 specifically for pathogen reduced plasma.
¶Reference ranges were taken from Erickson et al.18 and were established on fresh frozen, normal donor plasma samples by Machaon Diagnostic Laboratory
#The percentage of activity and the IU/mL unit are directly proportional (i.e., if the activity is 50%, it would be 0.5 IU/mL).
**Data from Heger et al.19
23. Can apheresis frozen plasma, psoralen-treated be used for alternative treatments?
Apheresis frozen plasma, psoralen-treated should not be used when coagulopathy can be more appropriately corrected with specific therapy, such as vitamin K or specific factor replacement. For example, replacement of the vitamin K-dependent coagulation factors (i.e., factors II, VII, IX and X) can be achieved with prothrombin complex concentrates.
Hemophilia A and B and von Willebrand disease are more appropriately treated with recombinant or virally inactivated fractionation products or 1-deamino-8-D-arginine vasopressin as initial treatment. For replacement of fibrinogen and factor XIII, commercial virally inactivated concentrates are also available. Some products are only available through the Special Access Programme of Health Canada.
Solvent detergent plasma is also available as an alternative to apheresis frozen plasma, psoralen-treated.